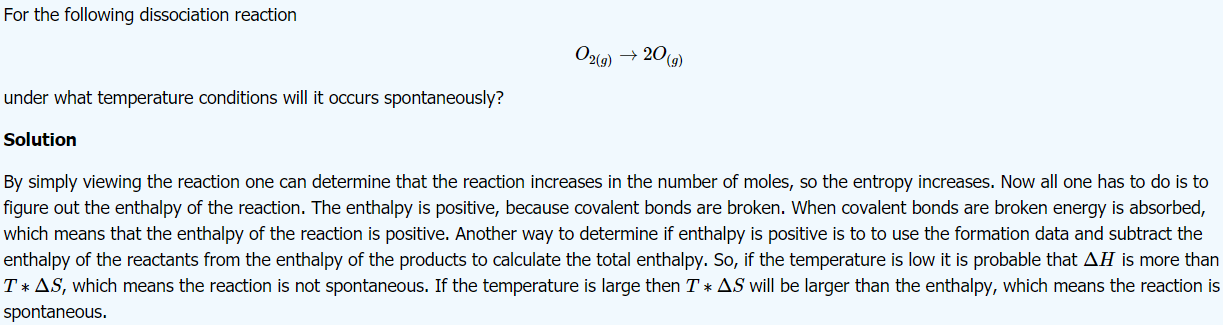SECOND LAW OF THERMODYNAMICS
Entropy(s)
 Entropy is a concept used to indicate the irregularity of a system. In other words, entropy is a measure of the irregularity of a system.
Entropy is a thermodynamic term that refers to thermal energy that cannot be translated into mechanical energy in a system.
Any event that occurs promptly and increases the irregularity of the system usually causes an increase in entropy.
However, the clear definition of entropy is as follows. "Heat energy that is not converted in a system is called entropy.".
In any physical and chemical event, useful energy is dispersed irregularly and imlessly. This energy is embossed by entropy.
The minimum entropy means that the energy loss is minimal, and the maximum entropy means that the entire energy will be lost.
Entropy is a concept used to indicate the irregularity of a system. In other words, entropy is a measure of the irregularity of a system.
Entropy is a thermodynamic term that refers to thermal energy that cannot be translated into mechanical energy in a system.
Any event that occurs promptly and increases the irregularity of the system usually causes an increase in entropy.
However, the clear definition of entropy is as follows. "Heat energy that is not converted in a system is called entropy.".
In any physical and chemical event, useful energy is dispersed irregularly and imlessly. This energy is embossed by entropy.
The minimum entropy means that the energy loss is minimal, and the maximum entropy means that the entire energy will be lost.
Entropy is a thermodynamic status function and is unlikely to be Ω
this happens in ; 
Here k Boltzman is constant. Entropy increases as heat is given to the system, entropy decreases as heat is taken from the system. Entropy exchange in a co-temperature process is given with this formula; 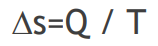 Note; Entropy is a measure of disorder.
Note; Entropy is a measure of disorder.
ΔS : amount of change in entropy.
ΔQ: change in heat energy.
T: absolute temperature.
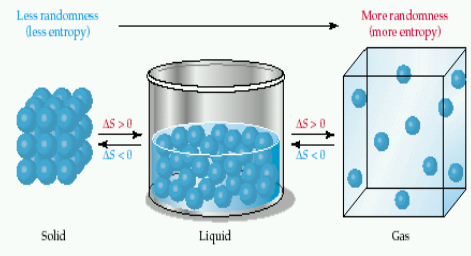
*The liquid state of a substance has higher entropy than its solid state.
*The gasely state of a substance has a higher entropy than its liquid state.
*As the temperature rises, the entropy of matter rises.
*As the number of gas molecules increases in a chemical reaction, entropy increases.
Important Physicists Involved In The Development Of The Second Law Of Thermodynamics.
1.
Nicolas Léonard Sadi Carnot is a French physicist known as the father of thermodynams. He noticed that some calories had disappeared in the motion cycle. Thus, the concept of thermodynamic reversialism proved to be wrong, and proved that reversal is the result of every system involving work.
2.
Rudolf Clausius was a German physicist and Clausius developed his expression. The second law of thermodynamics is expressed by Clausius as follows: It is impossible for a machine that works by performing a thermodynamic cycle to take heat from a low-temperature object and heat to a high-temperature object without any other energy interaction.
3.
William Thompson, also known as Lord Kelvin. Kelvin argued that it was impossible to convert heat completely. He formulated this situation. This means that it is impossible to turn it into work without losing some energy from all the energy of a system.
4.
Konstantin Karatodori, a Greek mathematician, has taken important steps for the second law.
Second Law
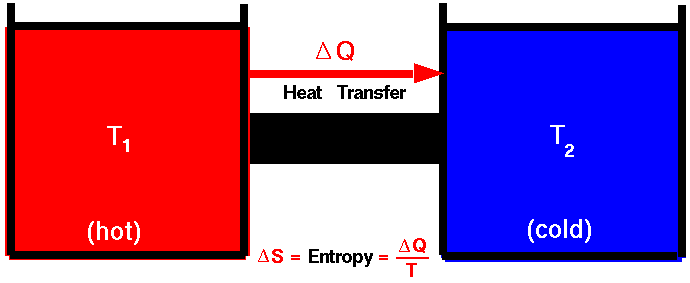
Heat transfer always flows from high temperature to low temperature. An insulated system prefers a state with maximum irregularity. This is also the case in which the probability is maximum. When an inseed system changes, the change in the system's entropy is greater than or zero than zero. It is not possible for a heat machine to turn thermal energy into work with 100% efficiency.
Any event that occurs promptly increases the entropy of the system. The change in entropy of a physical or chemical event that occurs in unseated systems is the sum of the change in the entropy of the system and the change in the entropy of the environment. Where the system is the environment in which the event occurs. This environment can be a container, laboratory or nature.
ΔS = ΔS (system) + ΔS (environment)
ΔS:Change in total entropy
ΔS (system):Change in the entropy of the system
ΔS (environment):Change in the entropy of the environment
As irregularity increases in the system, so increases entropy. This incident is related to Law II of thermodynams. For example, heat transfer is always from hot system to cold system. It is not possible to move the heat from the cold system to the hot system.
In a balanced event, it's rotating --> ΔS = 0
In a soli wouldn't be transformed. --> ΔS > 0
It's impossible to react in an inspicient reaction. --> ΔS < 0
ΔS is the entropy of the universe, also called total entropy. Total entropy is the sum of the entropys of the system in which the event occurred and the environment in which the system occurred.
Gibbs Free Energy
Gibbs Free Energy is used to directly express the entropy of a system. Free energy is energy ready to do business.
G=H-T.S
If a system is walking at constant temperature and pressure, this is the change in its free energy: ΔG = ΔH - T.ΔS
If reaction is prompt, ΔG < 0. In this case, the reaction reveals an energy that can do business.
For a reaction that occurs at constant temperature pressure,
If ΔG < 0, reaction is requested.
If ΔG > 0, the reaction is involuntary.
ΔG = 0 balances the reaction.
| Case | ΔH | ΔS | ΔG | Answer |
|---|---|---|---|---|
| high temperature | - | + | - | Spontaneous |
| low temperature | - | + | - | Spontaneous |
| high temperature | - | - | + | Nonspontaneous |
| low temperature | - | - | - | Spontaneous |
| high temperature | + | + | - | Spontaneous |
| low temperature | + | + | + | Nonspontaneous |
| high temperature | + | - | + | Nonspontaneous |
| low temperature | + | - | + | Nonspontaneous |