THERMODYNAMICS
Thermodynamics means heat (thermo) and power (dynamic). Thermodynamics is the science of heat, temperature and energy. Thermodynamics, which examines how energy in a system moves and converts into usable energy or heat, has emerged as a result of efforts to increase the efficiency of textile machines. Many technologies leading to the industrial revolution, for example the steam engine, have been developed thanks to their thermodynamic knowledge.
At the time of the industrial revolution, engineers were trying to build effective heat machines that do business with heat energy. To produce functional and efficient heat machines, basic research based on general principles has formed Thermodynamics which is one of the branches of physics. It is called studies on thermodynamics, heat and thermal energy. There are three laws of thermodynamics and in addition there is the fundamental law of zero.
In the early 18th century, it was thought that heat was a liquid called caloric, and that it flowed from hot objects to cold objects. In other words, when the calories in a cup of tea spread to the environment, the tea became cold and the cup and coffee table became hot. Benjamin Thompson discovered a flaw in the caloric theorem when examining where the heat generated during the puncture of a ball came from. The work done during the ball puncture caused heat to form. So heat could be created by doing a job. With this understanding of the relationship between heat and work and the beginning of mechanization, the concept of thermodynamics and the laws of thermodynamics have begun to emerge. Thermodynamics, which covers a wide range from daily goods to energy production systems, has 4 laws that still maintain their general validity.
INTRODUCTION
This law, which covers the relationship between heat and work, is an expression of the total amount of matter in the universe and the conservation of total energy. The first law of thermodynamics, the first law found in the most general form; He explains that 'energy cannot be destroyed when it is present, but can be transformed from one form to another'.
The 1st law is examined in two different ways for closed and open systems. In a system that is not related to its environment (closed system), the total energy is always the same. In systems with work and heat exchange between its environment (open system), the energy exchange is equal to the net heat exchange between the system and its environment.
.jpg)
In fact, this law states that substances and systems cannot heat themselves until a job is done. Einstein's special principle of relativity has shown that objects can be heated without a work input by exceeding this law. One of the best examples of this is that stars continue to burn for billions of years without any business input.
first Law of Thermodynamics
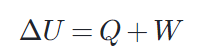
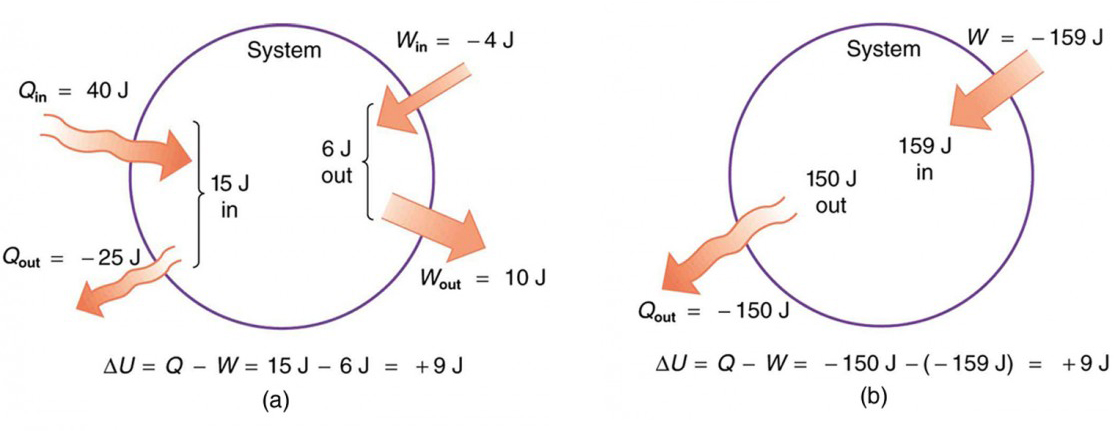
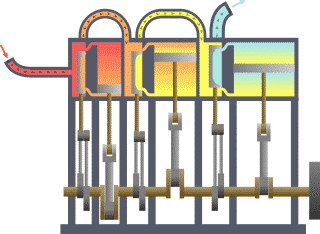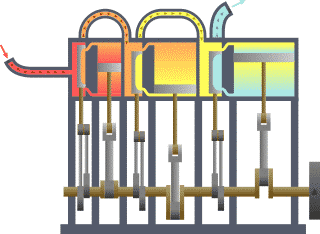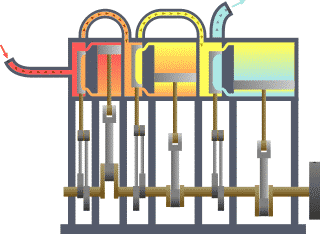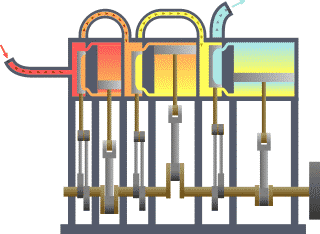
Many power plants and engines work by converting heat energy into work. This is because the heated gas is capable of operating and moving mechanical turbines or pistons. The first law of thermodynamics applies the principle of conservation of energy. Accordingly, heat transfer and the work performed by the system are among the methods of transferring energy into and out of the systems. According to the first law of thermodynamics, the internal energy ΔU of a system is the sum of the net heat transfer Q to the system and the net work (W) applied to the system. As the equation, the first law of thermodynamics is given as follows.
Here ΔU is the change in the internal energy U of the system. Q is the net heat transferred to the system, ie Q is the net sum of all heat transfers to and from the system. W is the net work done on the system.
That is, the heat with the surplus value Q adds energy to the system and the work with the surplus value W adds energy to the system. For this reason, the first law takes the form ΔU = Q + W. In other words, he says that we can add internal energy by heating or working on a system.
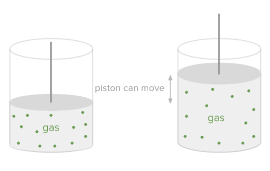
Nothing else is a better example of the first law of thermodynamics, except for gas (air or helium) in a mobile, but compact past piston. We will assume that the piston can move up and down, ie it can compress the gas or allow the gas to expand (but the gas cannot come out of the container).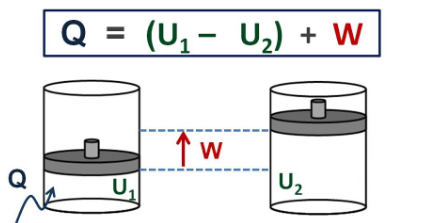 The gas molecules in the container are the "system". These gas molecules have kinetic energy.
The gas molecules in the container are the "system". These gas molecules have kinetic energy.
The internal energy U of our system can be considered as the sum of the kinetic energies of each gas molecule. That is, if the temperature T of the gas increases, the gas molecules are accelerated and the internal energy U increases (which means that ΔU is a plus sign). Similarly, if the temperature T of the gas decreases, the gas molecules slow down and the internal energy U of the gas decreases (which means that ΔU is minus-marked).
It is important to remember that when the speed of the gas molecules increases, both the internal energy (U) and the temperature (T) increase. Because there are two methods to measure the same thing, which is how much energy is in the system. Since the temperature and internal energy are proportional to each other, the temperature increases twice when the T∝U internal energy increases twice. Similarly, if the temperature does not change, the internal energy does not.
One way to increase the internal energy U (and thus its temperature) of the gas is to transfer heat (Q) to the gas. This can be done by placing the container on a Bunsen burner or by immersing it in boiling water. The high temperature environment will pass heat through the walls of the container to the gas and cause the gas molecules to move faster. If the heat enters the gas, Q will be marked plus. Conversely, we can reduce the internal energy of the gas by transferring heat from the gas. We can do this by putting the container in an ice bath. If heat is released from the gas, Q will be a minus sign. The marking rule for Q is explained as follows.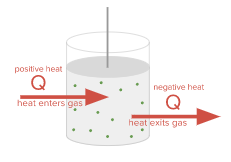
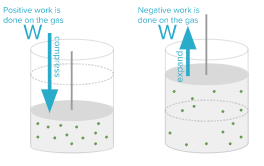
Since the pistons are moving, they can move downward and compress the gas and work on the gas. The collision of the downwardly moving piston and the gas molecules makes the gas molecules move faster and increases the total internal energy. If the gas is compressed, the work done on the gas becomes a plus sign. Conversely, if the gas expands and pushes the plunger upwards, it is the work done by the gas. Collision of the gas molecules with the retracted piston causes the gas molecules to slow down and reduce the internal energy of the gas. If the gas expands, the work done on the gas becomes a minus sign. The sign rule for W is given in the following figure.
↓↓↓The following table gives the signal rules for all three values mentioned above (ΔU, Q, W).↓↓↓
| ΔU (change in internal energy) | Q ( heat) | W (work done to gas) |
|---|---|---|
| Heat rises at T + | if the heat goes up to the gas + | gas is compressed + |
| If the temperature decreases T - | If the heat comes out of the gas - | If the gas expands - |
| if the temperature T is constant 0 | without heat exchange 0 | if volume is constant 0 |